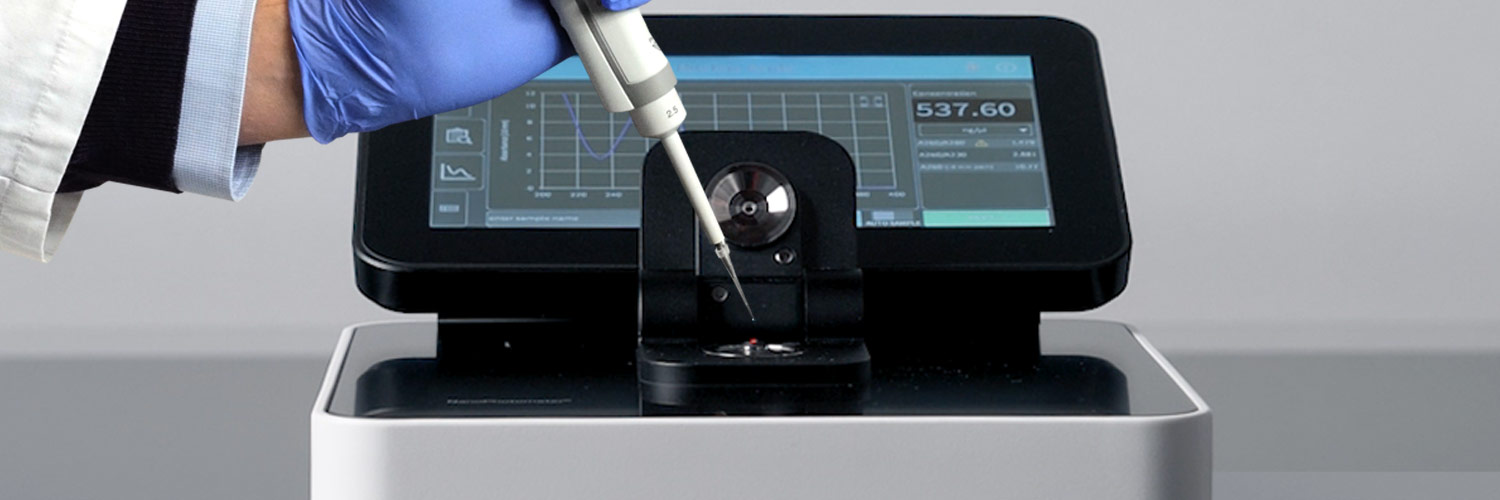



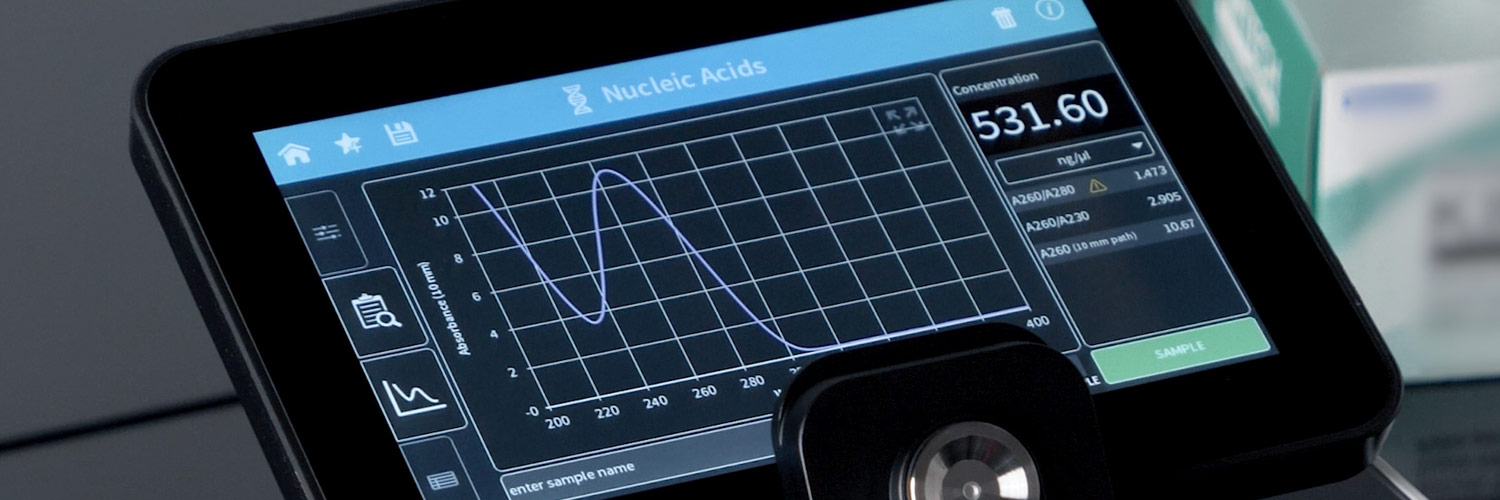
Implen NanoPhotometer®, Fully Compliant 21 CFR Part 11
Over the past years laboratories working in GMP / GLP / GxP regulated environments have been increasingly inquiring about the ability to add 21 CFR Part 11 compliance to their nanovolume UV/Vis spectrophotometer instruments. Since April 2019 eligible models from the Implen NanoPhotometer® product line can be upgraded with our CFR21 software package to help our users to become compliant with FDA requirements – Implen was again spearheading the market offering FDA 21CFR Part11 compliance for the NanoPhotometer® class of spectrophotometers – stand-alone with battery backup via the built-in computer and touch screen, through external computers or via Rest API with most LIMS systems.
Implen NanoPotometer® 21 CFR Part 11 Compliance
Fully Compliant!
- Limiting system access to authorized individuals
- Use of operational system checks
- Use of authority checks
- Use of device checks
- Determination that persons who develop, maintain, or use electronic systems have the education, training, and experience to perform their assigned tasks
- Establishment of and adherence to written policies that hold individuals accountable for actions initiated under their electronic signatures
- Appropriate controls over systems documentation
- Controls for open systems corresponding to controls for closed systems bulleted above
- Requirements related to electronic signatures
Ideal 21 CFR Tool for GLP/GMP/GxP Laboratories
- User management
- Access control
- Electronic signatures
- Data integrity
- Data security
- Audit trail functionality

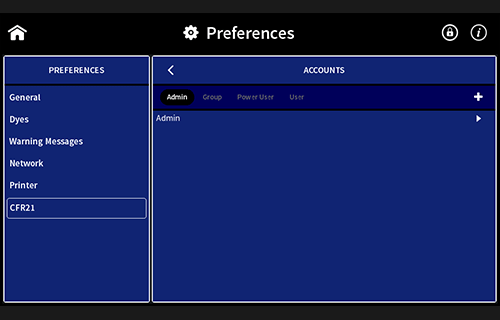
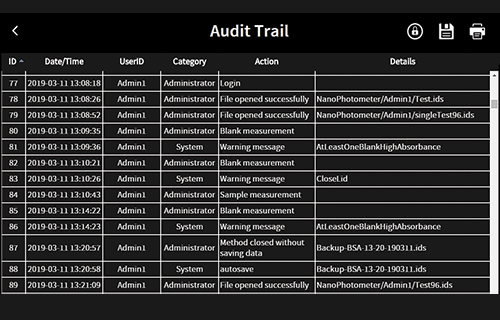
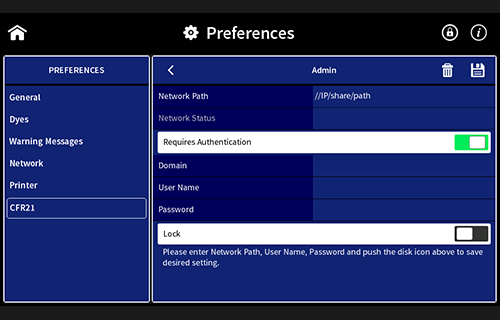
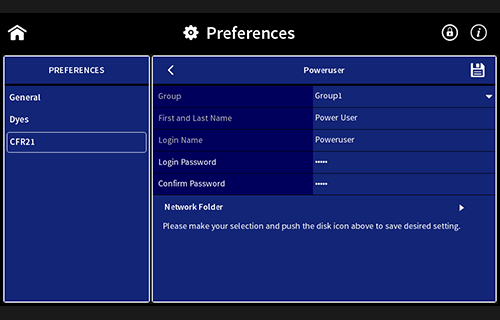
Installation and Operation Qualification (IQOQ)

LIMS Integration via REST API

Compare to Alternative Technologies
The table below demonstrates a comparison of the NanoDrop™ One/One C vs. our NanoPhotometer® line of spectrophotometers
| Features | NanoDrop™ One / One C | Implen NanoPhotometer® N60/NP80 |
| 21 CFR Part 11 Upgrade | ||
| Software Control | External control via Windows computer only | Stand-Alone and/or control via Windows and Mac OS |
| User Management | Yes | Yes |
| Audit Trail | Yes | Yes |
| Electronic Signature | Yes | Yes |
| Instrument Performance | ||
| Minimum Sample Volume | 1 µL 2 µl sample size is recommended for protein measurements | 0.3 – 2 μL |
| Pathlength | 0.03 – 1 mm Auto-ranging | Utilizes two fixed path lengths 0.67 and 0.07 mm Fixed path lengths never change over the lifetime of the instrument |
| Measurement Time | 8 seconds | 2.5 – 4 seconds (full scan 200 – 900 nm) |
| Enclosed Sample Measurement Surfaces | NO Column is exposed to surroundings | YES Enclosed sample measurement surfaces No evaporation Accurate results independent from environmental conditions Ability to measure proteins with detergent buffers without sample build-up |
| Photometric Range Cuvette | 0 – 1.5 A (with a 10 mm cuvette) | 0 – 2.6 A |
| Analyze Organic Solvent Samples in a Drop | Cuvette only | The NanoPhotometer® Sample Compression Technology allows the analysis of organic solvent samples in a drop and in a cuvette |
| Detection Range* | dsDNA: 2 – 27,500 ng/µl BSA: 0.06 – 820 mg/ml | dsDNA: 1 – 16,500 ng/µl BSA: 0.03 – 478 mg/ml |
| Optical Specifications | ||
| Measurement Wavelength | Full scan from 190 – 850 nm | Full scan from 200 – 900 nm |
| Illuminated Read Head | N/A | Illuminated read head for an easy sample application |
| Light Source | Xenon Flash Lamp | Xenon Flash Lamp |
| Instrument Features | ||
| Built-In Vortexter | N/A | YES 2,800 rpm; tube size up to 2.0 ml |
| Reconditioning-free | NO The pedestal of the instrument may become unconditioned over time, especially when measuring proteins or samples that contain surfactants or detergents. This requires reconditioning of the unit’s pedestal. | YES Scratch-resistant inert quartz pedestal that requires no reconditioning |
| Quality Control Warnings | Acclaro Technology with contaminant calculations | Sample Control™: quality control technology identifies sample impurities, potential contaminants, turbidity and lint residue Blank Control™: prevents inaccurate readings caused by high background blanks or insufficient cleaning |
| Built-in Web Application Server | NO Limited Android-based internal computer only | Full Web Application Server with REST API |
| Processing Power, Compatibility, Data Processing | ||
| Operating System | Android | Linux based NPOS (NanoPhotometer Operating System) Open platform to connect to Windows, Mac OS, Android, iOS via web application interface |
| Internal Data Storage | 32 GB | 32 GB for storage of thousands of measurements |
| Software Compatibility | Compatible with Windows only | Compatible with Windows 7, 8, 10 (32 and 64 bit), Mac OS X, iOS and Android OS |
| Data Transfer and Connectivity Options | The unit includes 3x USB A, Ethernet, Bluetooth, WiFi | The unit includes 2x USB A, USB B, HDMI, Ethernet, WiFi |
| Data Output | Separate as .csv .tsv .sql | Complete data as PDF & Excel |
| Instrument Control | The device can be connected to a computer via Ethernet cable for operation, control and data storage. | Easy control of the NanoPhotometer is possible via Wi-Fi from tablets or smartphones (Android OS & iOS). Control from PCs via LAN, WiFi or USB (Windows & Mac). The HDMI port allows for presentations directly from the device. |
| WiFi Hotspot | Client possible | Built-in WiFi hotspot and with WiFi client capability |
| Instrument Dimensions | 20 × 25.4 × 32.3 cm (8 × 10 × 12.7 inches) | 20 x 20 x 12 cm (7.9 x 7.9 x 4.7 inches) |
The table below demonstrates a comparison of the spectrophotometers 8-channel NanoDrop™ 8000 vs. 12-channel NanoPhotometer® N120
| Features | NanoDrop™ 8000 | Implen NanoPhotometer® N120 |
| 21 CFR Part 11 Upgrade | ||
| Software Control | N/A | Stand-Alone and/or control via Windows and Mac OS |
| User Management | N/A | Yes |
| Audit Trail | N/A | Yes |
| Electronic Signature | N/A | Yes |
| Instrument Performance | ||
| Number of Samples | Up to 8 | Up to 12 Single sample mode available |
| Minimum Sample Volume | 1 µL 2 µl sample size is recommended for protein measurements | 2 – 3.5 μL |
| Pathlength | 0.2 and 1 mm | Utilizes two fixed path lengths 1 and 0.125 mm Fixed path lengths never change over the lifetime of the instrument |
| Measurement Time | Less than 20 seconds 220 – 750 seconds for 8 samples | 20 seconds for 12 samples (full scan 200 – 900 nm) |
| Enclosed Sample Measurement Surfaces | NO Column is exposed to surroundings | YES Enclosed sample measurement surfaces No evaporation Accurate results independent from environmental conditions Ability to measure proteins with detergent buffers without sample build-up |
| Analyze Organic Solvent Samples in a Drop | N/A | The NanoPhotometer® Sample Compression Technology allows the analysis of organic solvent samples in a drop and in a cuvette |
| Detection Range* | dsDNA: 2.5 – 3,700 ng/µl BSA: 0.15 – 100 mg/ml |
dsDNA: 2 – 8,000 ng/µl BSA: 0.06 – 230 mg/ml |
| Optical Specifications | ||
| Measurement Wavelength | Full scan from 190 – 850 nm | Full scan from 200 – 900 nm |
| Illuminated Read Head | N/A | Illuminated read head for an easy sample application |
| Light Source | Xenon Flash Lamp | Xenon Flash Lamp |
| Instrument Features | ||
| Quality Control Warnings | N/A | Sample Control™: quality control technology identifies sample impurities, potential contaminants, turbidity and lint residue Blank Control™: prevents inaccurate readings caused by high background blanks or insufficient cleaning |
| Built-in Web Application Server | N/A | Full Web Application Server with REST API |
| Processing Power, Compatibility, Data Processing | ||
| Operating System | N/A Needs to be connected to a Windows computer |
Linux based NPOS (NanoPhotometer Operating System) Open platform to connect to Windows, Mac OS, Android, iOS via web application interface |
| Onboard Processor | N/A | Built-in computer with high-performance Intel Celeron dual core 2.4 GHz processor |
| Internal Data Storage | N/A | 128 GB for storage of thousands of measurements |
| Software Compatibility | Compatible with Windows only |
Compatible with Windows 7, 8, 10 (32 and 64 bit), Mac OS X, iOS and Android OS |
| Data Transfer and Connectivity Options | USB | The unit includes 2x USB A, USB B, HDMI, Ethernet, WiFi |
| Data Output | Separate as .csv .tsv .sql | Complete data as PDF & Excel |
| Instrument Control | The device must be connected to a computer via USB cable for operation, control and data storage. | Easy control of the NanoPhotometer is possible via Wi-Fi from tablets or smartphones (Android OS & iOS). Control from PCs via LAN, WiFi or USB (Windows & Mac). The HDMI port allows for presentations directly from the device. |
| WiFi Hotspot | N/A | Built-in WiFi hotspot and with WiFi client capability |
| Instrument Dimensions | 24 × 32 cm (89.4 × 12.6 inches) |
20 x 20 x 12 cm (7.9 x 7.9 x 4.7 inches) |
* dsDNA tends to precipitate/coagulate at concentrations above 10,000 ng/µl and should be diluted.
NanoDrop™ and NanoDrop™ One are registered trademarks of Thermo Fisher Scientific.
Implen® is not affiliated, sponsored or endorsed by Thermo Fisher Scientific or NanoDrop™.
Implen’s Overcoming 21 CFR Part 11 Limitations of Other Technologies
Have you experienced any of these difficulties finding the right technology?
- No user management/21 CFR part 11 compliant software available
- User management/21 CFR part 11 compliant software only available via external computer (no stand alone option)
- No REST API interface available to connect to your LIMS System
- Complex and pricy IQOQ procedures only offered with an expensive contract for onsite service
- Long measurement times
- Cumbersome software user interface
- Costly recalibration/maintenance or decreasing accuracy over time
- Instrument’s service and support discontinued and software not compatible with Windows10 or Mac OS
- Low reproducibility (mainly caused by evaporation effects)
- Getting results for more than 40 samples per run is time consuming
- Waited for weeks to get instrument repaired
How about trying a device that overcomes all of these 21 CFR issues?

Implen NanoPhotometers® Reviews
“Excellent design and is easy to use“
Rating: 5.0 ★★★★★
Application Area: Neurosciences
“The photometer has excellent design, and is easy to use. Built-in vortex is one of the smartest decisions I’ve seen in lab equipment. Data manipulaton and interface decrease hand-on time, and are user-friendly.”
Olga Starkina
Organization: Melsytech LLC, Russia
“Really impressed with the accuracy, ease of use, and increased sample throughput“
Rating: 5.0 ★★★★★
Application Area: DNA Analysis
“I’ve really been impressed with the accuracy, ease of use, and increased sample throughput with this instrument. All this in such a small and sleek black box. The touch screen interface is a nice feature and very straightforward. You can easily navigate through the applications, load templates, access and analyze your data. I like that you’re given the option to run up to 12 samples at a time but you can also choose to run 1 or 2 samples, customizable to your experiments. Having options is always a plus and you get that with the N120. The pipette guide makes it really easy to apply your samples. I can see that a lot of time, expertise, and careful considerations went into developing the NanoPhotometer.”
Nam Che
Organization: UCLA Department of Medicine, USA
“Accurate Measurements, Solid Device“
Rating: 5.0 ★★★★★
Application Area: DNA Concentration Quantification
“My lab does a lot of molecular cloning work for synthetic biology projects. For the longest time we didn’t have any reliable and simple way of quantifying DNA concentration. This device has been a a life saver. It is accurate (checked samples against other similar devices), easy to use, has lots of extra features that the competitors lack and is cheaper.
If you are just needing to quantify DNA concentration the N50 is a solid choice. But it comes with so many other features you’ll be left wondering how other manufacturers get away with charging more for less. Overall the device is very sturdy and produces accurate results. But beyond just the basics you can do protein and kinetics assays and more. The touchscreen is easy an intuitive. Results are saved directly to the machine as a pdf or spreadsheet and can be retrieved remotely by a tablet/phone or a desktop computer. I prefer to simply copy the pdf directly to my laptop over the network. Another great feature is that the sample pedestal has a visible light shining through it while you load the sample which makes it easier to see what you’re doing. That is something I have not seen on any other microvolume spectrophotometer. My only complaint, if I have to come up with one, is that typing on the touchscreen with my arm extended out in front of my is kind of clumsy. But its a minor inconvenience. I just stick to short samples names.”
Cory Tobin
Organization: TheLab Inc, USA
“Best small volume spec on the market“
Rating: 5.0 ★★★★★
Application Area: Protein/nucleic acid quantitation
“The Implen NanoPhotometer N50 is extremely easy to use (intuitive menus and settings), accurate, and genuinely capable of measuring very small volumes repeatably. The touch screen works well even with gloves. Window configurations are adaptable so you can customize the information you’re looking at while working. The instrument is light and mobile, and since it’s all-in-one, you can literally move it to where you’re working as needed. It’s not stuck in a particular corner of the lab. In my opinion, it’s the best nano-scale measurement device on the market for routine lab sample quantification and spectral reading.”
David Rawling
Organization: Inflammatix, Inc., USA
“Easy use and accurate results “
Rating: 4.7 ★★★★★
“We are using it for nucleic acid quantification. It’s so easy to use, gives accurate results and easy to save data in organized way. Would recommend it.”
Dr. Assala Samad
Organization: University of Oxford, United Kingdom
#1 Alternatives from Implen
NanoDrop™ and NanoDrop™ One are registered trademarks of Thermo Fisher Scientific.
Implen® is not affiliated, sponsored or endorsed by Thermo Fisher Scientific or NanoDrop™.